高氯酸锂,Lithium perchlorate;anhydrous, ≥99.9% trace metals basis
最近浏览商品
-
![显示明细 白细胞酯酶,Leukocyte esterase [LE];≥20 units/mg protein(biuret) 图片 白细胞酯酶,Leukocyte esterase [LE];≥20 units/mg protein(biuret)](https://www.bszh.cn/images/thumbs/0015029_leukocyte-esterase-le20-unitsmg-proteinbiuret_32.jpeg) 白细胞酯酶,Leukocyte esterase [LE];≥20 units/mg protein(biuret)
白细胞酯酶,Leukocyte esterase [LE];≥20 units/mg protein(biuret) -
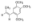 β-细辛脑,β-Asarone;phyproof® Reference Substance, ≥90.0% (HPLC)
β-细辛脑,β-Asarone;phyproof® Reference Substance, ≥90.0% (HPLC) -
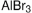 溴化铝,Aluminum bromide;anhydrous, powder, 99.999% trace metals basis
溴化铝,Aluminum bromide;anhydrous, powder, 99.999% trace metals basis -
![显示明细 伊红美蓝琼脂 [EMB],Eosin-Methylene Blue Agar 图片 伊红美蓝琼脂 [EMB],Eosin-Methylene Blue Agar](https://www.bszh.cn/images/thumbs/default-image_32.png) 伊红美蓝琼脂 [EMB],Eosin-Methylene Blue Agar
伊红美蓝琼脂 [EMB],Eosin-Methylene Blue Agar -
 甲基-PEG12-叠氮化物,Methyl-PEG12-Azide;≥98%
甲基-PEG12-叠氮化物,Methyl-PEG12-Azide;≥98% -
 正己烷,Hexane;HPLC Plus, for HPLC, GC, and residue analysis, ≥95%
正己烷,Hexane;HPLC Plus, for HPLC, GC, and residue analysis, ≥95%

